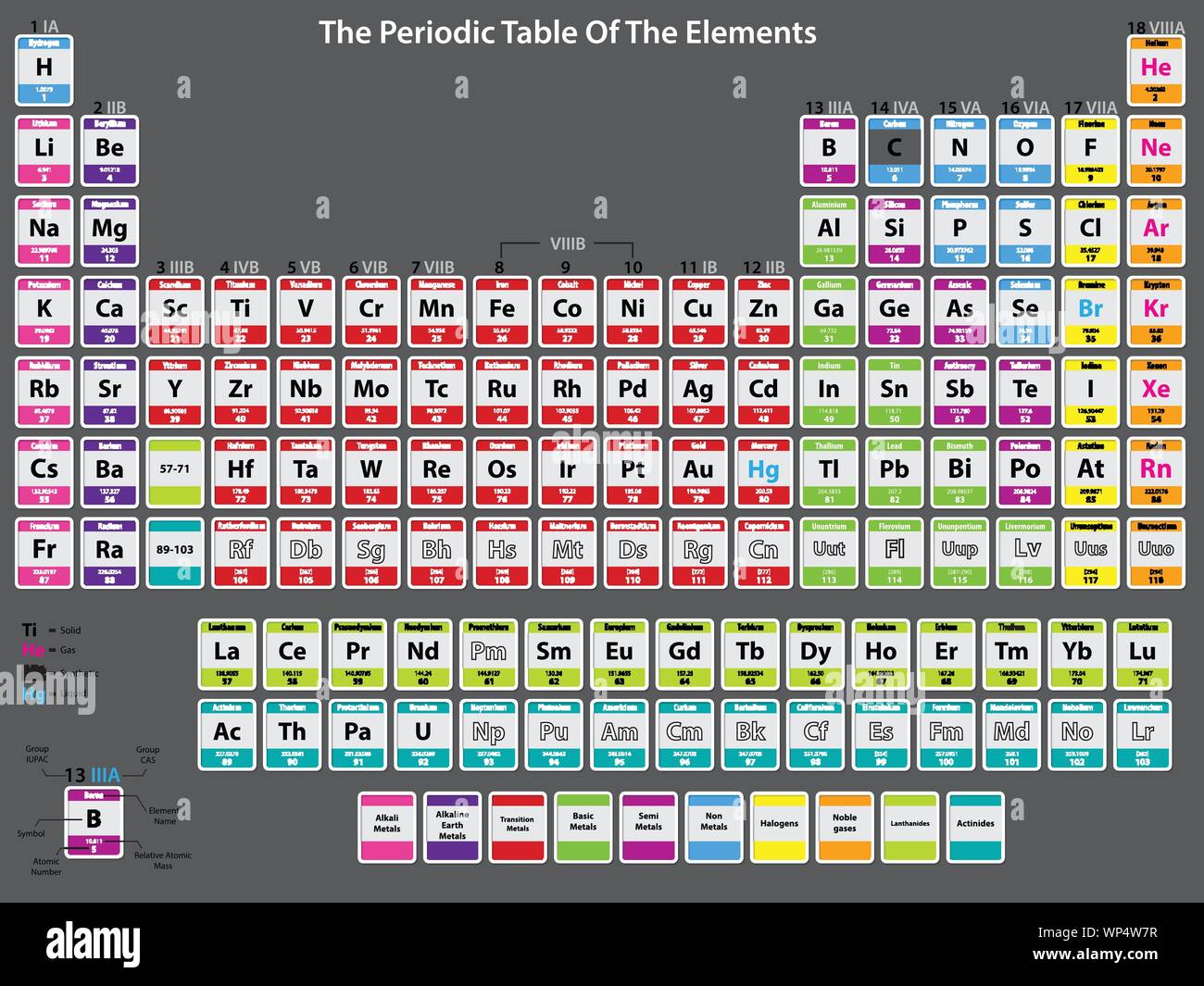
Radon is the only radioactive element from group 18. These elements occur as monatomic species and exist as gases under room-temperature. Although 209Bi holds the half-life record for alpha decay, bismuth does not have the longest half-life of any radionuclide to be found experimentally-this distinction belongs to tellurium-128 (128Te) with a half-life estimated at 7.7 × 1024years by double β-decay (beta decay).The nonmetallic elements categorized under group 18 – helium, neon, argon, krypton, xenon, and radon – are called noble gases. The decay event produces a 3.14 MeV alpha particle and converts the atom to thallium-205.ĭue to its extraordinarily long half-life, for nearly all applications 209Bi can still be treated as if it were non-radioactive. Theory had previously predicted a half-life of 4.6×1019 years. Wow, I just looked up Bismuth, incredibleīismuth-209 was long thought to have the heaviest stable nucleus of any element, but in 2003, a research team at the Institut d’Astrophysique Spatiale in Orsay, France, discovered that 209Bi undergoes alpha decaywith a half-life of approximately 19 exayears(1.9×1019 which is 19 quintillion years), over a billion times longer than the current estimated age of the universe. I think checking out some educational YouTube videos would help you. If lead does decay into something else, it would probably take us billions of years to find out, right? Only specific configurations of protons and neutrons are stable, everything else is unstable and undergoes radioactive decay. Isotopes are atoms with the same number of protons but different numbers of neutrons. I’ve already told you that every single element has radioactive isotopes, see: - note just how many radioactive isotopes lead has, many are decay products from other elements, lead is the heaviest element that still has stable isotopes. When stars explode a large amount of heavier elements are formed, but again this isn’t the same process.Ĭould lead ever decay into something else? Is there a half-life of lead? What makes your ask this question? Helium will never “decay” into lead, nor will neon etc. Oh cool, so does that mean that, like radon, all of the noble elements will eventually turn into lead? Radon is a fairly bad building block for life because it isn't very useful to all the reactions required for what we imagien "life" to be. Radon, being inert and very unreactive, would essentially be trying to get milk from a rock. It reacts to a lot of things in a lot of very useful ways. The reason carbon based lifeforms are so succsessful is because carbon is really versatile. While I can't say it's impossible it's unlikely. It will get darker, colder, stars will die out one by one, and then an eternity of nothing.Ĭould there eventually be a life-form made of radon Assuming you're not religious, the world has no goal. 
Noble gases already are happy, so they don't feel a need to interact with other elements. Those two elements might chemically interact, one giving away two electrons to the other so both are happy. Other elements might have 6 electrons when they Really want to have 8. They neither want to give away orbitals nor add any, they are content. Without going too much in to chemistry they have properties that make them more likely to be inert: f.i they have a decent number of electrons in their orbitals. Noble elements aren't "special" in the sense that they are more stable than other chemicals. What do they say about critics? That you need them to make an ironclad and attack-proof argument? At the very least you helped me take the temperature of reddit's opinions which are actually normally even grosser and more leftistĭoes the presence of noble gases imply tbat eventually that all the elements of the world will eventually become "noble" elements? *Conclusion: I think we've reached moot points but I thank you all for being patient with me (if witg occasionally frustrated and hostile "patience.") I think even though we disagreed for large chunks of the discussion, it seems like you still helped me learn something I don't really know much at all about the guy Walter Greiner so idk about his stance, no clue if it's right or not Gross opinions on the subject by Feynman and Pauli: Could we find up to 200 elements? Up to 500 elements? 7,000 elements? 2 million elements? Infinite elements? To use an example from the simplest noble gas- hydrogen decays into helium, will helium ever decay into something else? As a response to an earlier comment, will helium or radon or neon ever decay? If not, in that sense, will the whole universe eventually be noble gases?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
